Our Team at MAGHREB PHARMA Expo 2023
-

Netraja
Associate Director , Sales and Marketing - Turkey , Africa
-

Karun Gaur
Vice President - SEMEA
-

Namrata
Vice President - HR
About MAGHREB PHARMA Expo 2023
MAGHREB PHARMA Expo 2023 is the premier African pharmaceutical industry event. For the past 9 years, the expo has established itself as the go-to platform for pharmaceutical industry professionals in the region.

The expo brings together a diverse range of exhibitors showcasing the latest innovations in the field including pharmaceutical ingredients, cutting-edge production lines, advanced packaging solutions, state-of-the-art clean rooms, treatment systems for air, water production, sterile lines, biotech reactors, laboratory equipment, and quality control technologies.
With over 2,600 decision-makers in attendance from Algeria, Morocco, Tunisia, Mali, Mauritania, Libya, and beyond, the expo provides a unique opportunity for exhibitors to connect with key players in the industry and showcase their products and services to a highly engaged and qualified audience.
In addition to the exhibition, the expo also features a comprehensive conference program, including "learnshop" sessions where industry experts will be sharing their insights and knowledge on the latest developments in the pharmaceutical industry. These sessions are designed to provide valuable information and offer opportunities to network with other professionals in the field.
We look forward to meeting you at MAGHREB PHARMA Expo 2023, an event that is sure to inspire, inform and engage industry professionals. Come and be a part of the conversation shaping the future of the pharmaceutical industry in Africa.
About Dr.Reddy’s API Business
We specialise in manufacturing and supplying a wide range of APIs for generic formulation manufacturers in India and overseas. The company has a diversified portfolio of 150+ active pharmaceutical ingredients, which are used in major therapeutic areas such as gastrointestinal, cardiovascular, diabetology, oncology, pain management, and dermatology. Our API manufacturing facilities adhere to cGMP (ICH Q7) and are inspected on a regular basis by international regulatory bodies. We have eight commercially inspected USFDA production plants, six in India and one each in Mexico and the United Kingdom.
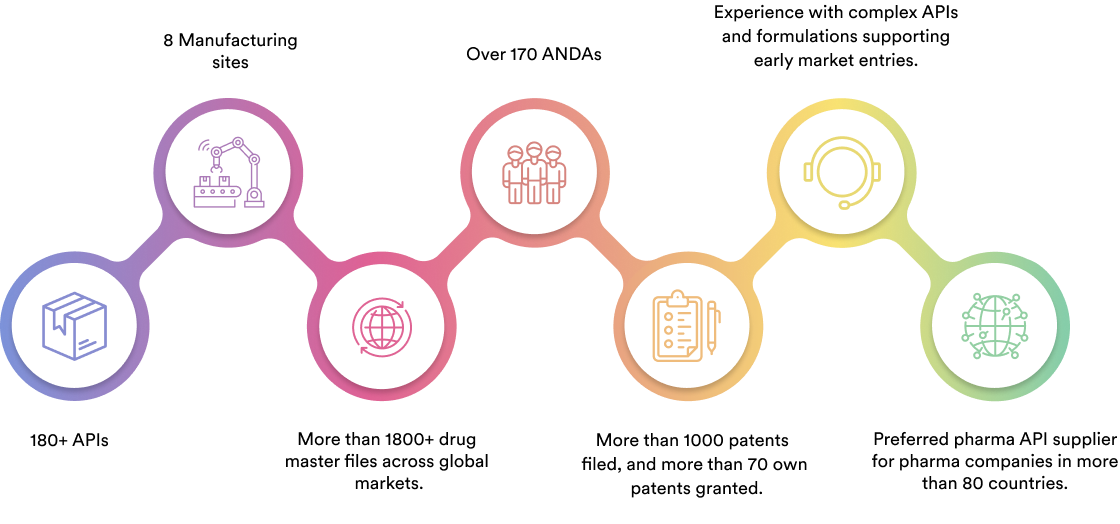
Dr. Reddy's API business operates in a number of global markets, including the United States of America, Latin America, Europe, India, Russia, and other CIS nations.
Sustainability
Dr. Reddy's is among the top sustainable pharmaceutical companies globally. We are listed in the sustainability yearbook of S&P 2022 for the second year in a row and the Bloomberg Gender-Equality Index (GEI) for the fifth consecutive year. In addition, we were ranked ninth in the Dow Jones Sustainability Index (DJSI) 2021 among the most sustainable pharmaceutical companies in the world. In our sustainability journey, these accolades are humbling and demonstrate that we're on the right track. These awards recognize our consistent performance in the environment, social, and governance (ESG) framework. Sustainability and ESG are becoming increasingly important topics for all stakeholders, so we will strive to maintain visibility in these areas.

Our Achievements

07/10/2021
The winner of the Best Indian Company in the US - Manufacturing Sector for the year 2021


03/11/2020
Dr. Reddy’s Laboratories wins “API Supplier of the Year” award at the Global Generics & Biosimilars Awards 2020

Our plants

免責事項
このウェブサイト上のいかなる情報も、製品またはサービスへの言及を含め、販売の申し出を構成するものではなく、販売の申し出を表すものと解釈されるものでもありません。有効な特許により保護されている製品は、商用目的で提供または提供されるものではありません。ただし、特定のケースでは、Dr. Reddy の独自の裁量により、現地の法的要件に従って、そのような製品の研究用数量が、規制免除が存在する場所に、インド特許法第 107A 条 (Bolar 免除) に基づく規制提出の目的で提供される場合があります。購入者は、それぞれの市場における特許シナリオを含む製品またはサービスについて独自の評価を行う必要があり、すべての特許関連法的責任を負うことになります。Dr. Reddy は、商品性、特定目的への適合性、および非侵害の保証を含むがこれらに限定されない、明示または黙示を問わずすべての保証を否認します。

