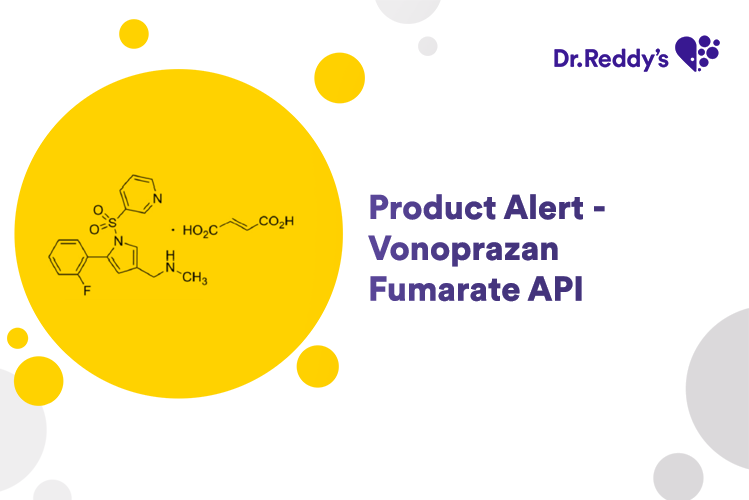
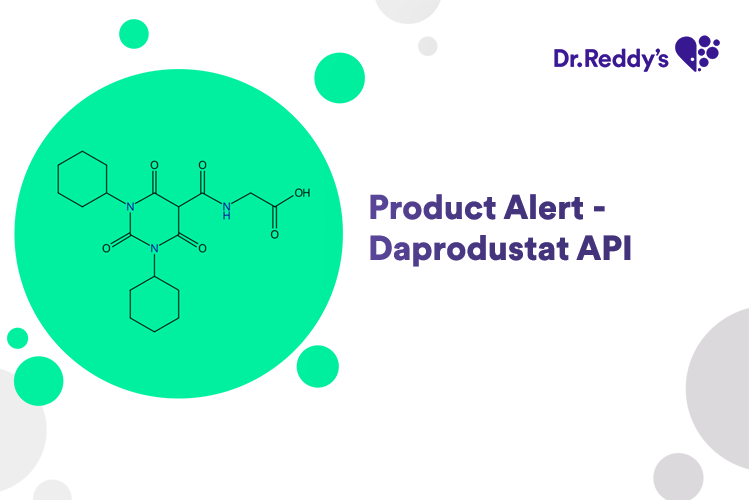
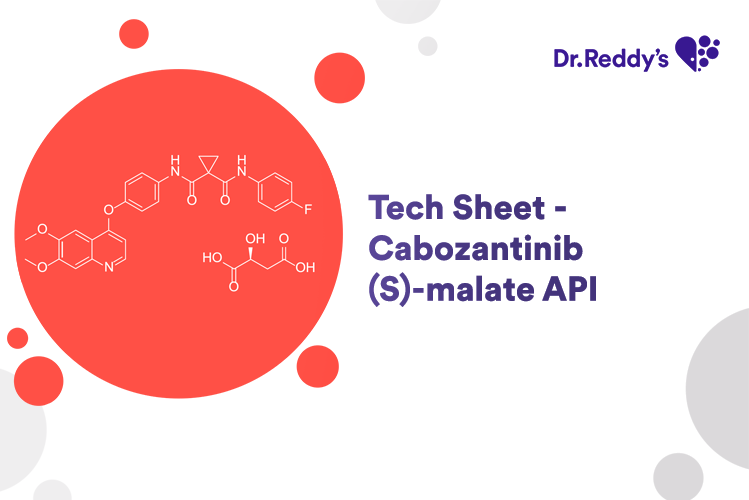
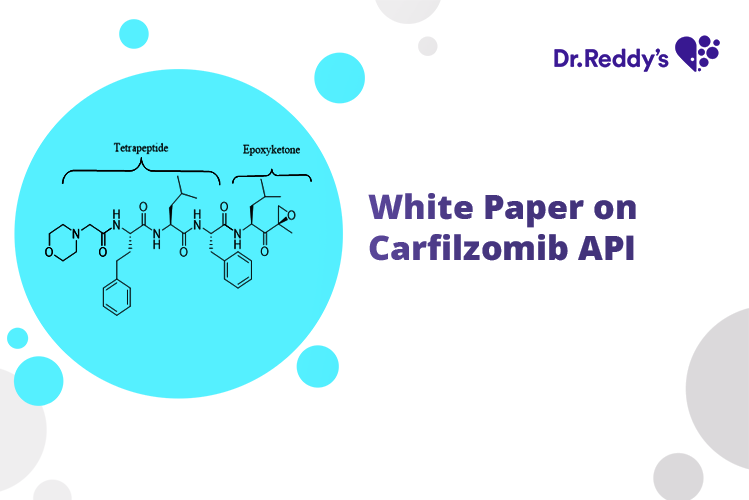
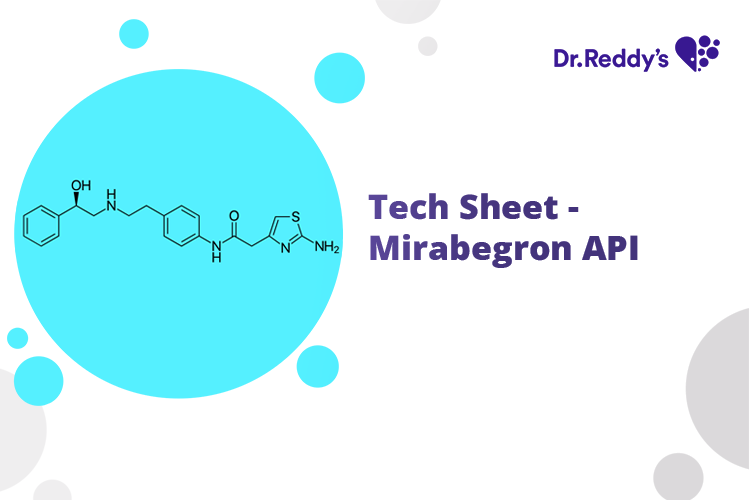
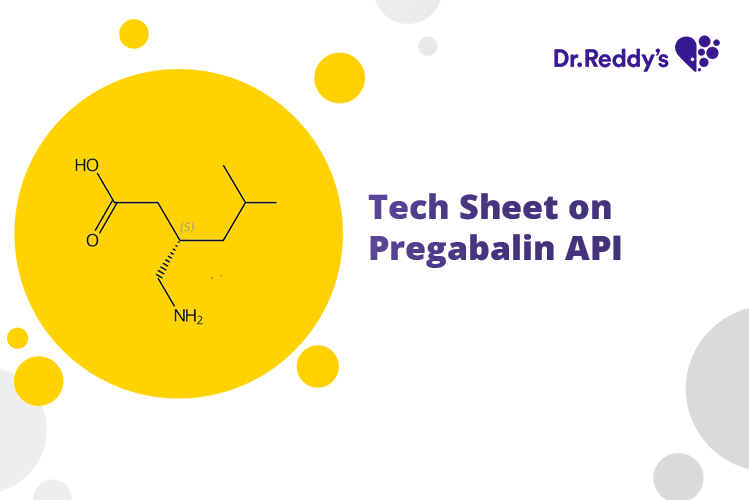
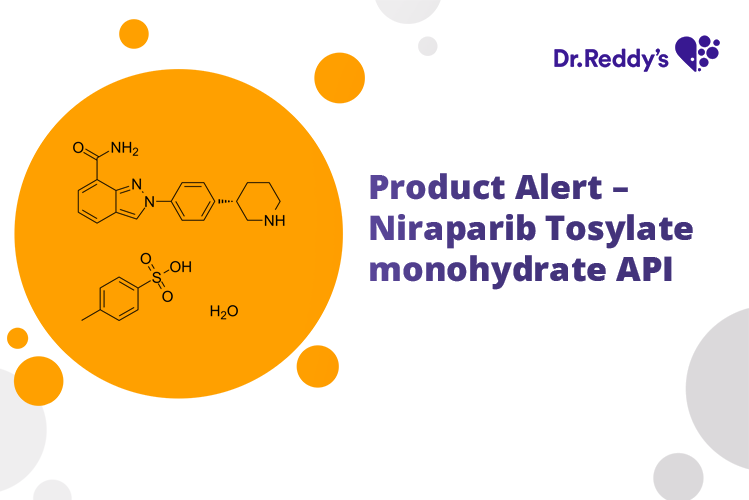
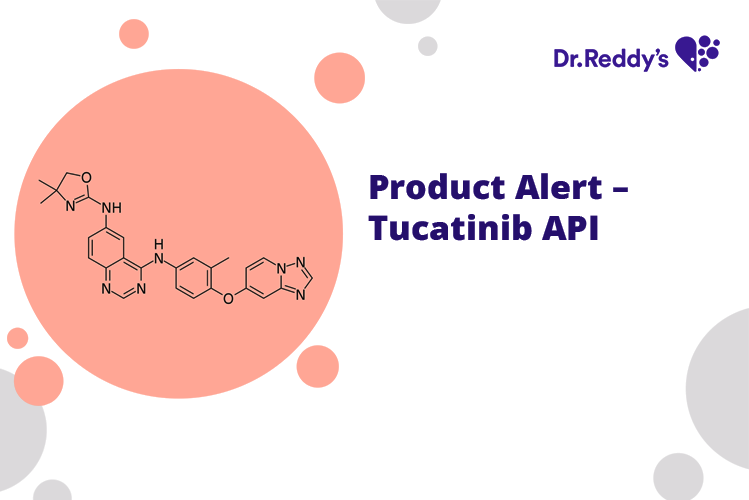
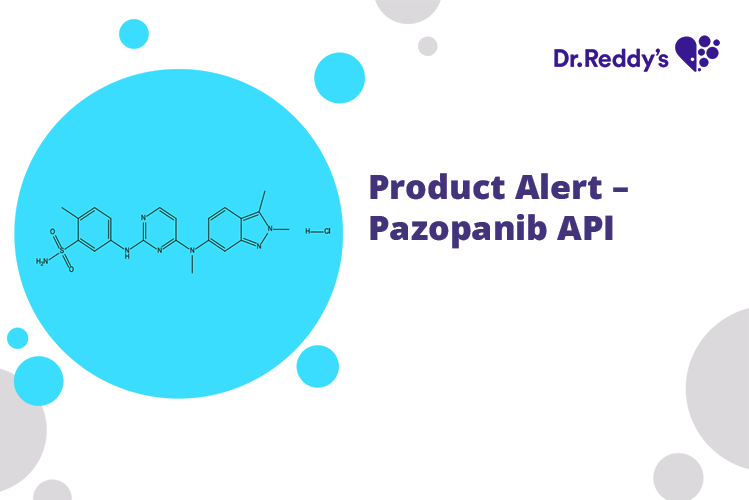
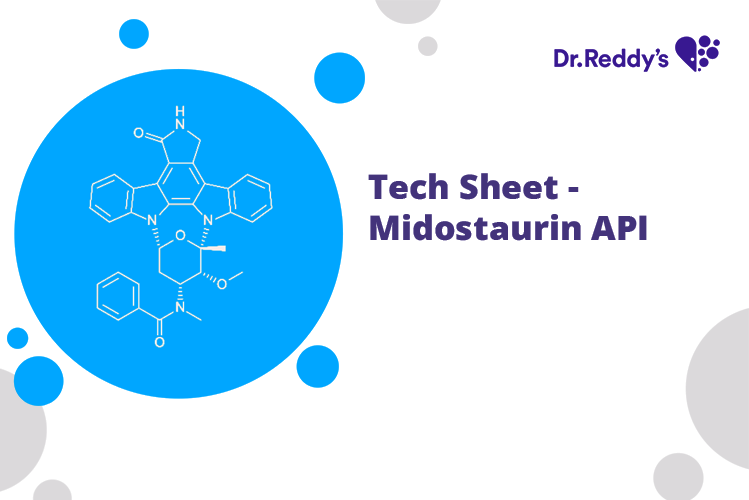
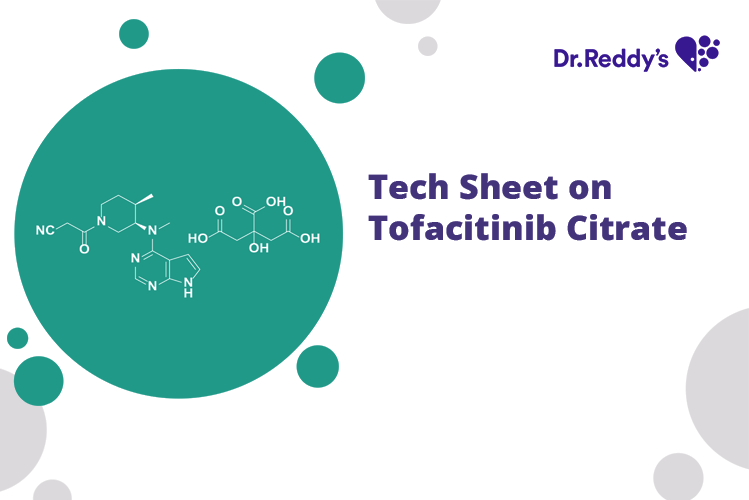
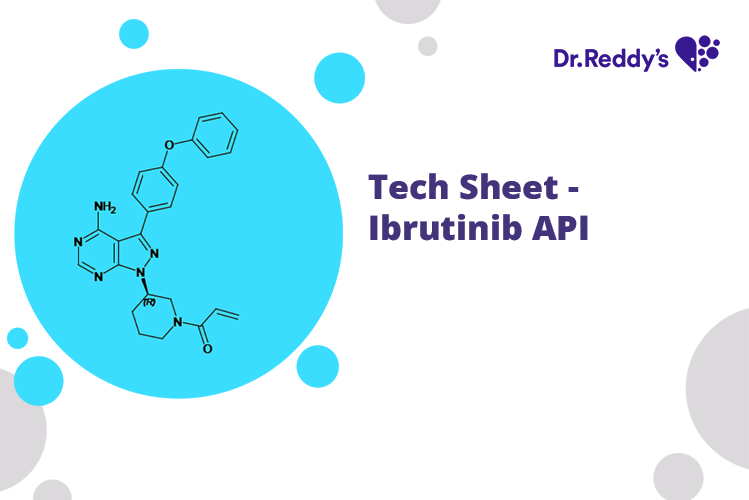
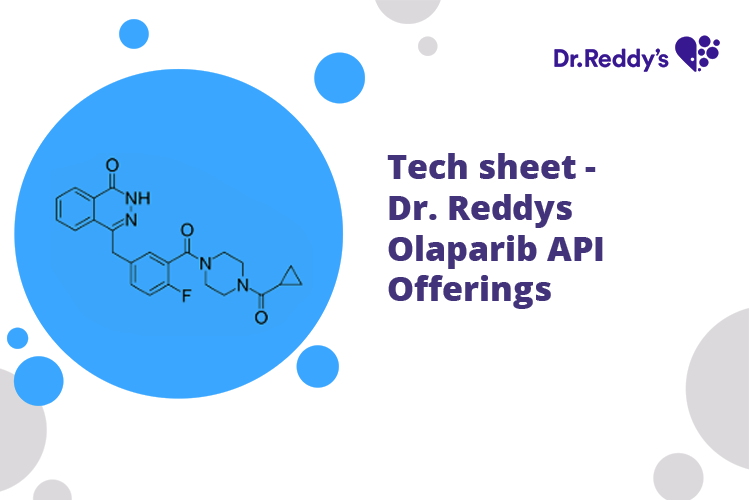
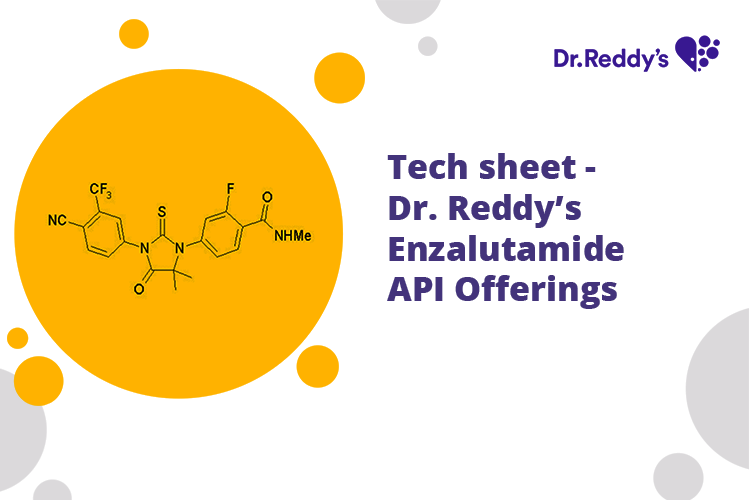
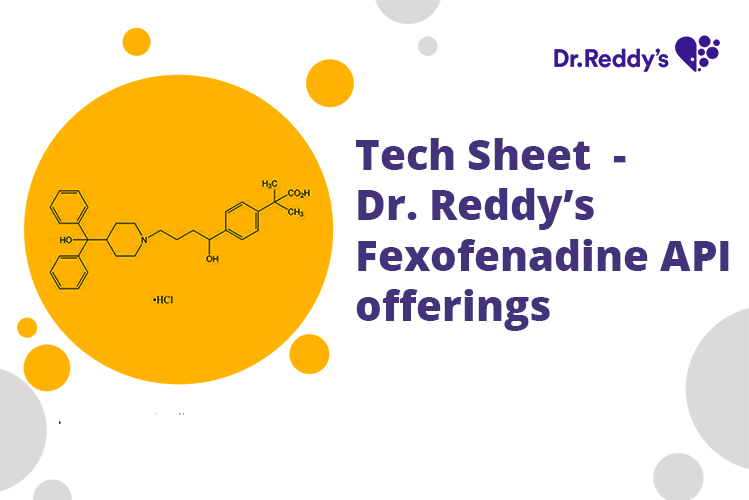
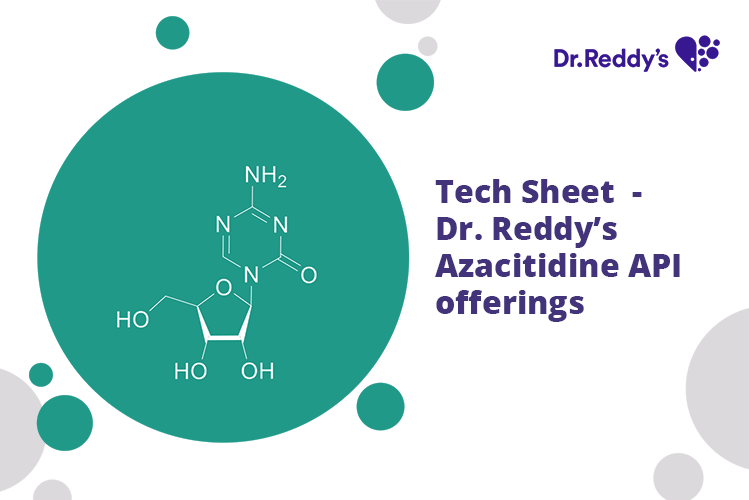
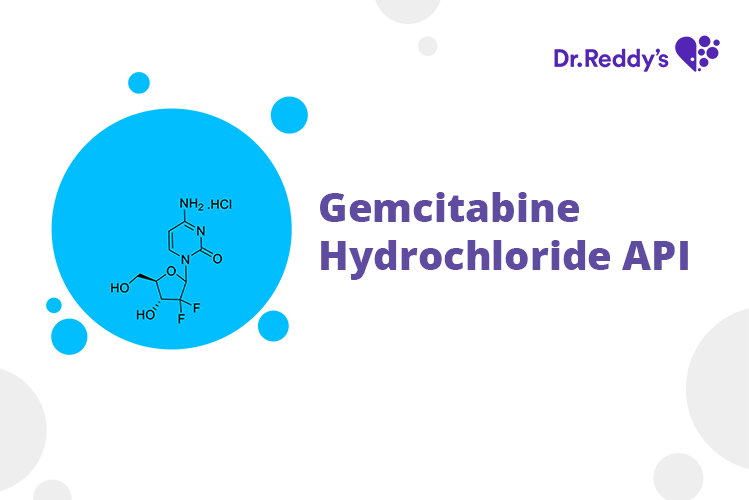
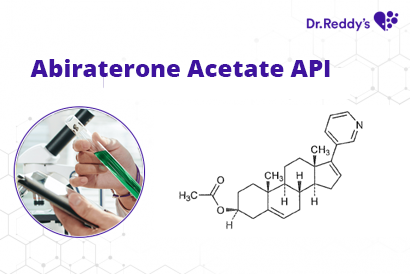
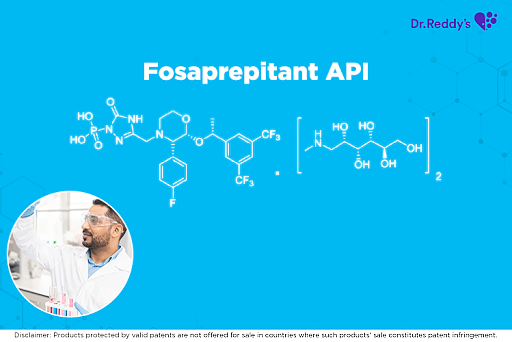
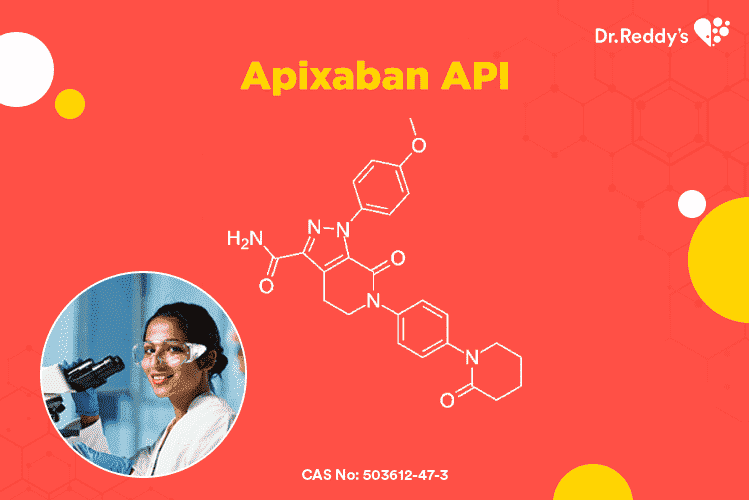
Meet with our product experts in one-on-one virtual sessions
Set Up a MeetingDisclaimer
No information in this catalog - including any reference to any product or service - constitutes an offer for sale, or be construed as representing an offer for sale. Products protected under valid patents are not offered or supplied for commercial use. However, the research quantities of such products may be offered for the purpose of regulatory submissions, wherever such regulatory exemptions exist. The buyers should make their independent evaluation of the patent scenario for their respective markets and will be responsible for all patent related liabilities. Products protected under valid patents in India are not available for commercial use but would be available for Section 107A purposes.
API Linkedin
page yet?
Follow this page to explore how our offerings can accelerate your formulation success