Our Team at CPHI China 2026
About Dr.Reddy's API Business
We specialise in manufacturing and supplying a wide range of APIs for generic formulation manufacturers in India and overseas. The company has a diversified portfolio of 250+ active pharmaceutical ingredients, which are used in major therapeutic areas such as gastrointestinal, cardiovascular, diabetology, oncology, pain management, and dermatology. Our API manufacturing facilities adhere to cGMP (ICH Q7) and are inspected on a regular basis by international regulatory bodies. We have eight commercially inspected USFDA production plants, six in India and one each in Mexico and the United Kingdom.
Dr. Reddy’s API business operates in a number of global markets, including the United States of America, Latin America, Europe, India, Russia, and other CIS nations.

250+ APIs

8 Manufacturing sites

2000+ drug master files across global markets.

Over 325 ANDAs

Filed 1100+ patents filed globally

Experience with complex APIs and formulations supporting early market entries

Preferred pharma API supplier for pharma companies in more than 80 countries

Sustainability is deeply embedded in our purpose and forms the core of our organization.
Our journey began with APIs in 1984, and we evolved from small molecules to large molecules and from APIs to finished formulations over the decades. Amidst this evolution, what has remained constant at the heart of our organization is a deep commitment to patients and the customers we serve through our core principles of collaboration and sustainability. By the end of FY2025, we had served over 756 million patients and continue our endeavours to reach over 1.5 billion patients globally by 2030. This means almost one in every five people on this planet will use Dr. Reddy’s product or service. The health of people is strongly linked to the health of our planet. That's why our integrated report also shows how sustainability and business are strongly linked.
Our ESG (Environment, Social & Governance) Goal:
Building the future responsibly
Our ESG goals are deeply embedded in our business strategy, guiding our efforts to create a significant and lasting positive impact on the planet and its people. We have made fair progress across several of these goals, even as a few continue to present challenges. We are committed to advancing our ESG journey, ensuring that as we prosper, we do so responsibly and with resilience.
What we achieved in the past year:
3.8
Bn total revenue (17% growth)
165
New products launched
111
DMFs filed, (1,630+ Cumulative DMFs)
249
Dossiers, including 10 ANDAs filed
76
Filings in the US pending for approval
20.4%
Women in leadership
96%
Employees trained for skill upgradation
0.14 to 0.07
*LTIFR reduced from
126
Differently abled employees
700,000+
Lives impacted through CSR
783
Million KL of water saved through agri-water saving techniques
66,723
MTCO2e of direct emissions reduction
11%
Reduction in water intensity per rupee of turnover with respect to FY2024
60%
Carbon neutrality achieved
75,761
KL of harvested rainwater used
54%
Water reused/recycled
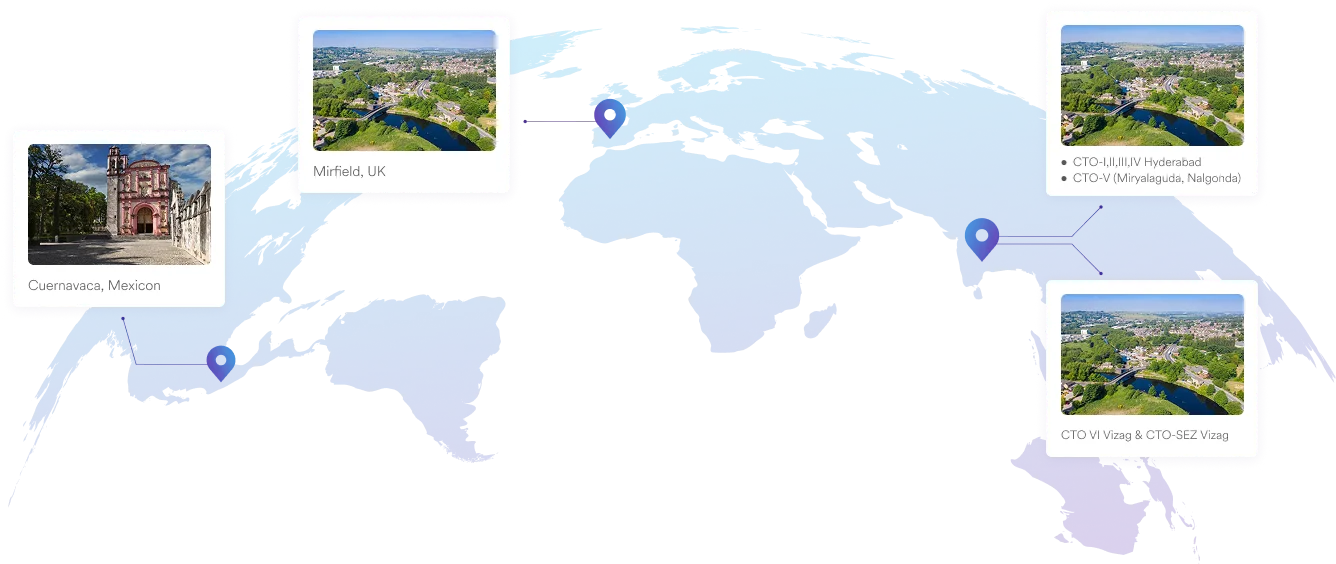
Our Plants

What We Present at CPHI China 2026
APIs for Global Markets, Including China
- A strong and diversified API portfolio across major therapeutic areas:
- Cardiovascular, CNS, Gastro‑Intestinal, Anti‑infectives, Oncology, Dermatology, and Metabolic disorders
- APIs with China DMF, USDMF, and EU DMF coverage
- Complex APIs and value‑added technologies
- Pipeline APIs aligned with future market and regulatory needs
- Support for both commercial supply and development programs
Finished Dosage Forms (FDFs)
- We showcase our formulation and manufacturing capabilities across:
- Oral solid dosage forms, injectables, and complex generics
- R&D‑driven formulation expertise
- Supply partnerships, in‑licensing, and out‑licensing opportunities
- End‑to‑end support from development to commercialization
Peptides Capabilities:
- cGMP manufacturing supported by robust quality systems
- Flexible and scalable production, from grams to multi‑kg
- A strong global regulatory track record
- Collaboration opportunities for peptide development through Aurigene and Dr. Reddy’s
- Support for clinical, development, and commercial supply
CDMO & Custom Services
- Comprehensive, end‑to‑end development and manufacturing services
- API development and scale‑up
- Peptide synthesis and manufacturing
- Small‑molecule CDMO services
- Custom synthesis and process optimization
- Seamless clinical‑to‑commercial supply
Biosimilars
- We present our global biosimilar portfolio with partnership opportunities for China and international markets:
- Commercial and late‑stage biosimilars
- Flexible licensing and partnership models
- High‑quality, affordable biologics supported by strong clinical data
B2B Collaborations
- Co‑development opportunities
- In‑licensing and out‑licensing
- Strategic sourcing and long‑term supply agreements
- Technology transfer and R&D collaborations
Our Commitment
- Across APIs, FDFs, Peptides, CDMO, and Biosimilars
- Quality and regulatory excellence
- Reliable and consistent supply
- Sustainable and compliant manufacturing
- Supporting customers with future‑ready pharmaceutical solutions












