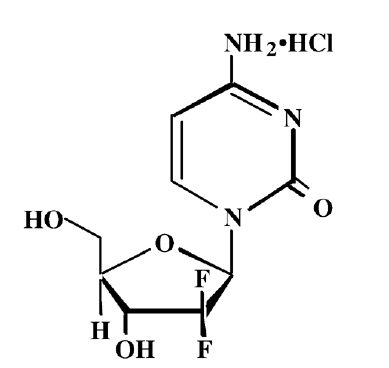
盐酸吉西他滨 API
CAS Number: 122111-03-9
About 盐酸吉西他滨 API
Therapeutic Category
Anti-Cancer/ Oncology
API Technology
High Potent
Dose Form
Injectable
Dr Reddy's Development Status
Available
Available Regulatory Filing
China DMF, Brazil DMF
作用机制
吉西他滨(Gemcitabine)可杀死正在进行DNA合成的细胞,阻断细胞通过G1/ s期边界的进程吉西他滨可通过核苷激酶代谢为二磷酸(dFdCDP)和三磷酸(dFdCTP)核苷。 吉西他滨二磷酸可抑制核糖核苷酸还原酶,这是一种负责催化生成脱氧核苷三磷酸用于DNA合成反应的酶,导致脱氧核苷酸浓度的降低,包括dCTP。 吉西他滨三磷酸与dCTP竞争并入DNA。 通过二磷酸作用降低dCTP细胞内浓度,从而增强吉西他滨三磷酸与DNA的结合(自我增强作用)。 吉西他滨核苷酸并入DNA后,只有一个额外的核苷酸被添加到不断增长的DNA链中,这最终导致凋亡细胞死亡的开始。
适应症
Gemzar®是一种核苷代谢抑制剂,适用于:
- 与卡铂合用于铂基治疗完成后至少6个月后复发的晚期卵巢癌的治疗。.
- 与紫杉醇合用于既往含蒽环类辅助化疗失败后转移性乳腺癌的一线治疗,除非蒽环类药物是临床禁用的。
- 与顺铂合用治疗非小细胞肺癌。
- 作为治疗胰腺癌的单一药物。
Dr. Reddy's的专长
Dr. Reddy's总部位于印度海得拉巴,是全球领先的活性药物成分(API)供应商之一。 Dr. Reddy's的API业务是美国、欧洲、巴西、拉丁美洲、日本、中国、韩国和新兴市场的制药公司的首选合作伙伴。
Dr. Reddy's博士的API业务在过去30多年来在开发和制造复杂API(如类固醇,多肽,复杂长链分子和高效API(HPAPI /肿瘤药物))方面所建立的深厚技术优势中茁壮成长。 我们在知识产权和法规事务方面的实力可以帮助我们始终如一地达到并超越监管标准,从而为这一专业知识提供补充。 Dr. Reddy's博士 盐酸吉西他滨 API是研发,知识产权和监管方面广泛专业知识的结果。
帮助我们的客户率先进入市场的关键组成部分是响应式供应链。 我们通过确保所有设施都高效运行并达到最新的质量,安全和生产率标准来实现这一目标。 业务和工厂之间的强大互联可以快速响应动态的市场变化,从而避免短缺并满足需求的突然激增。
免责声明
本目錄中的任何信息(包括對任何產品或服務的任何引用)均不構成銷售要約,或被解釋為代表銷售要約。受有效專利保護的產品不提供或供應用於商業用途。但是,只要存在此類監管豁免,就可以出於監管提交的目的提供此類產品的研究數量。買方應對各自市場的專利方案進行獨立評估,並承擔所有與專利相關的責任。在印度受有效專利保護的產品不可用於商業用途,但可用於第 107A 節。
FAQs
Preacautions should be taken for unusual bleeding or bruising, black, tarry stools, blood in the urine or stools, or pinpoint red spots on your skin.
Gemicitabine is used to treat breast cancer that cannot be treated with or that did not respond to chemotherapy that included an anthracycline drug.
Gemcitabine Hydrochloride is a BCS class III drug of choice in the treatment of cancer, as a single or in combination chemotherapy.
Gemcitabine have DMF filling in the US, Brazil, CEP, Europe, Canada, Japan, Korea and Russia.
Gemcitabine Hydrochloride should be stored at 2° to 8°C (36° to 46°F).
Gemcitabine is available in dosage form of EQ 200 MG BASE/Vial
Insights Delivered
Sign-up for our email service to get Market and Product insights and updates right to your digital doorstep
The categories of personal information collected in this form include name, company, and contact information etc. The personal information collected will be used for exploratory discussions on contract manufacturing, marketing and to perform research and analytics and others. For more information about the categories of personal information collected by Dr.Reddy's and the purposes for which Dr.Reddy's uses personal information, visit https://api.drreddys.com/privacy-policy.
免责声明
本網站上的任何信息,包括對任何產品或服務的任何提及,均不構成銷售要約或被解釋為代表銷售要約。受有效專利保護的產品不得提供或供應用於商業用途。但是,在某些情況下,雷迪博士可自行決定並根據當地法律要求,在存在此類監管豁免的任何地方,提供此類產品的研究數量,以根據《印度專利法》第 107A 條(Bolar 豁免)進行監管提交。購買者應對其各自市場的產品或服務(包括專利情況)進行獨立評估,並對所有與專利相關的責任負責。 Dr. Reddy's 不承擔任何明示或暗示的保證,包括但不限於適銷性、適用於特定用途和非侵權的保證。



